World
Military horses that broke free in London in serious condition
Technology
Fallout 4 Next-Gen Update Rolling Out Now
Emerson College protests broken up by Boston police, 108 arrested at pro-Palestinian camp, 4 officers hurt
U.S.
BOSTON – Boston Police arrested more than 100 people as they cleared out pro-Palestinian protesters and their encampment from Emerson College early …
How Taylor Swift Writes About Being Taylor Swift
Entertainment
The real main character in Swift’s latest album, ‘The Tortured Poets Department’ isn’t an ex, but Swift herself. How she’s chosen to …
SNP’s power-sharing deal with the Scottish Greens collapses
World
By Mary McCool & Craig Williams BBC Scotland News 25 April 2024, 07:36 BST Updated 51 minutes ago To play this content, …
Ford looks to future EV breakthroughs — and smaller cars — to staunch the bleeding
Business
Ford is the No. 2 seller of electric vehicles in the US. It’s very proud of that fact, but the amount of …
One UI 6.1.1 could be focused on video AI innovation
Technology
Last updated: April 25th, 2024 at 16:57 UTC+02:00 Samsung appears fully committed to its new AI strategy for mobile devices, TVs, and …
Ex-Coyotes receive emotional welcome in Salt Lake City: ‘We already want to fight for Utah’
Sports
SALT LAKE CITY — The door to the chartered Delta Airlines plane carrying the inaugural members of Utah’s NHL team popped open …
Hubble image may contain evidence of stellar cannibalism in dumbbell-shaped nebula
Science
Sign up for CNN’s Wonder Theory science newsletter. Explore the universe with news on fascinating discoveries, scientific advancements and more. CNN — The …
Zoonotic Transfer Suspected After Two Human Deaths : ScienceAlert
Health
A medical case report suggests that a deadly prion disease may have made its way from deer into humans. Two hunters have …





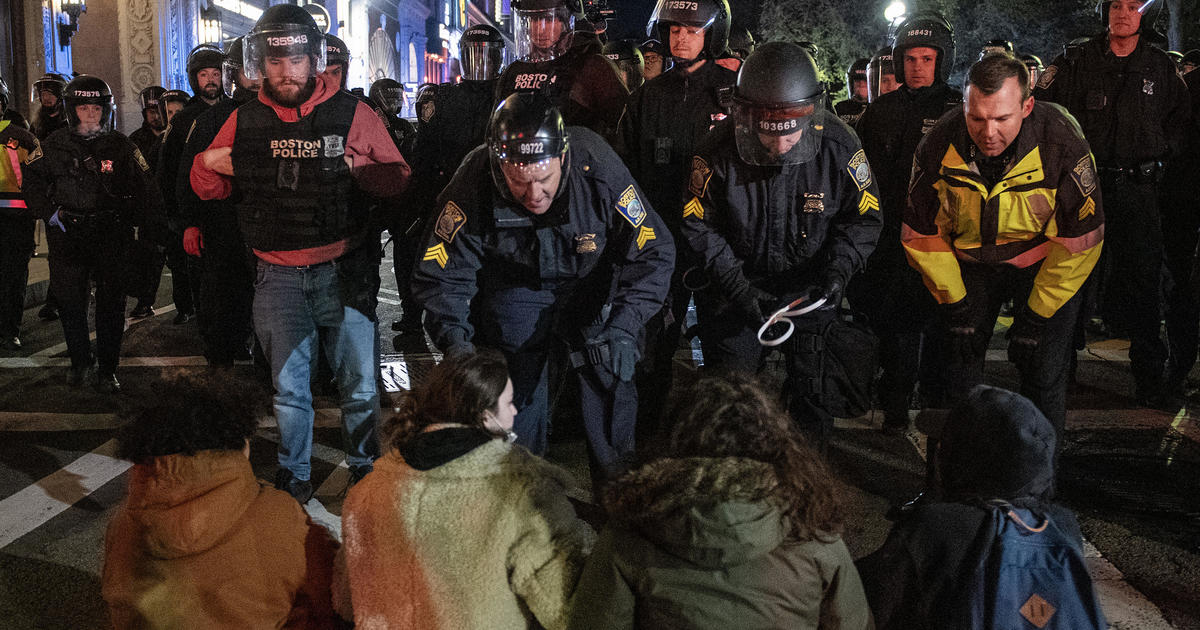
:no_upscale()/cdn.vox-cdn.com/uploads/chorus_image/image/73304940/TaylorOnTaylor_Getty_Ringer.0.jpg)

/cdn.vox-cdn.com/uploads/chorus_asset/file/24917756/2024_ford_f_150_stx_exterior_106_64ff7327a5c0f.jpg)